Context
Seabed exploration and exploitation represent a growing challenge for many industries, requiring the implementation of corrosion protection methods suited to deep environments. Cathodic protection, commonly used for steel structures in marine environments, must be sized from reliable data characterising the metal/seawater interface. Yet many parameters influence corrosion and cathodic protection current demand — temperature, dissolved oxygen content, biological activity, hydrostatic pressure, formation of calcareous deposits — and few experimental data are available for depths greater than 500 m. Moreover, results obtained in the laboratory or in surface waters cannot be directly extrapolated to great depths, which underlines the need for long-term in situ field data.
Facilities
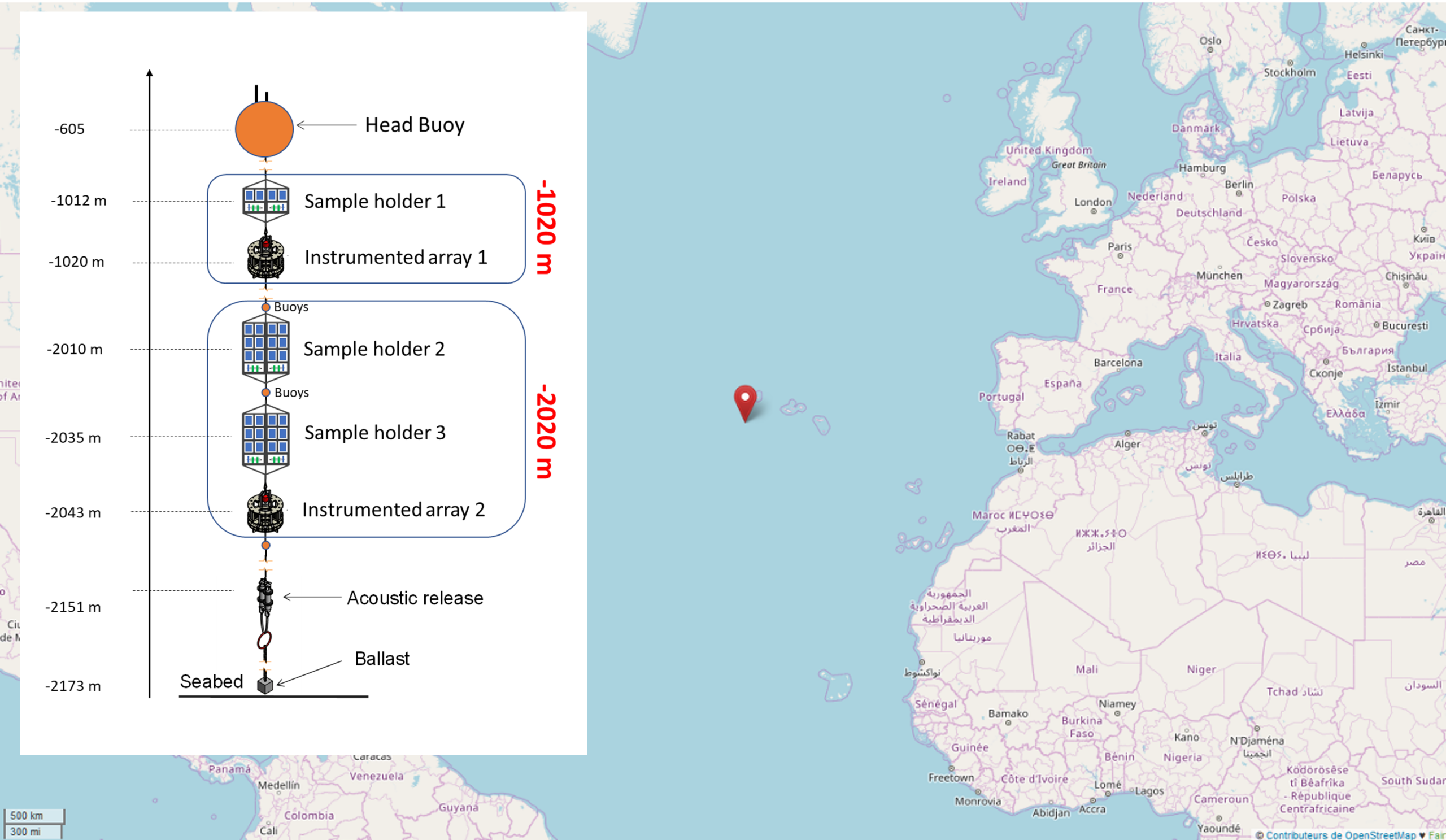
The study was based on the deployment of an instrumented mooring line for 11 months in the Atlantic Ocean (Azores region), at two depths: an intermediate depth (1,020 m) and a great depth (2,020 m). Carbon steel corrosion coupons were exposed at each depth for the determination of corrosion rates by mass loss. In situ cathodic protection sensors (SCPCs) coupling a carbon steel cathode with aluminium-indium (AlIn) and aluminium-gallium (AlGa) galvanic anodes, with different shunt resistors, enabled pseudo-polarisation curves to be built at various exposure times and the capacities of the galvanic anodes to be assessed. Environmental parameters (temperature, dissolved oxygen, current velocity, salinity) were recorded continuously throughout the exposure. The calcareous deposits formed on the specimens were characterised after recovery by Raman spectroscopy, scanning electron microscopy coupled with energy-dispersive analysis (SEM/EDX), and X-ray diffraction (XRD).
Results
The corrosion rates measured in intermediate and deep waters are comparable and remain within the expected range for carbon steel in quiescent seawater, with the slightly higher value in intermediate water attributed to higher temperature and greater oxygen diffusion. The cathodic protection current densities measured after 11 months are consistent with the literature and decrease over time due to the gradual formation of a calcareous deposit, this decrease being more pronounced in deep water, suggesting the formation of a more protective deposit. Characterisation of the calcareous deposits reveals that in cold deep water, calcite and monohydrocalcite are favoured over aragonite, that the Ca/Mg ratio decreases with depth, and that mixed calcium-magnesium compounds appear under the effect of the high hydrostatic pressure. Finally, the measured capacities of the AlIn and AlGa galvanic anodes are consistent with the literature data for long-term exposures.