Context
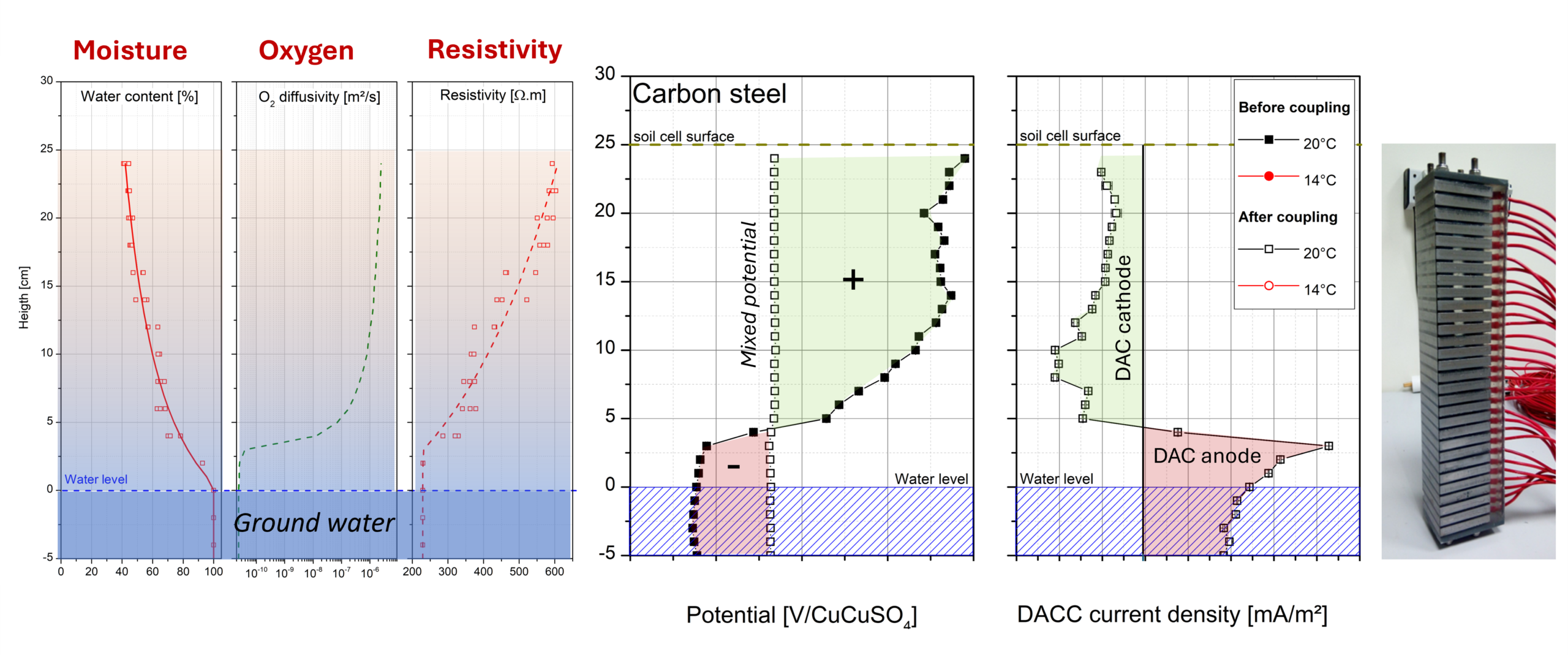
Soil is a complex porous medium whose physico-chemical properties, in particular water content, strongly condition the corrosion mechanisms of buried metallic structures. Close to the water table or at the interface between two soil horizons, a moisture gradient may develop, inducing local heterogeneity in oxygen access. This phenomenon gives rise to differential aeration corrosion cells (DACCs), which redistribute anodic and cathodic processes along the metallic structure through galvanic coupling. These cells are particularly problematic for large linear buried structures such as pipelines, tubular piles or sheet piles, where a portion of the structure may simultaneously be in soil zones of very different aeration. Experimental quantification of this phenomenon, in particular for galvanised steel-type materials, had been poorly documented in the literature.
Facilities
The study is based on two complementary approaches. Firstly, a specific DACC module consisting of 30 specimens of carbon steel or electrogalvanised steel (50×50×8 mm) stacked and separated by an insulating epoxy resin was exposed in two types of soil subjected to a vertical moisture gradient, reconstituted by capillarity from a bottom filled with deionised water. This setup allows individual measurement of the corrosion potentials and galvanic coupling currents before and after electrical connection of the specimens. Secondly, a one-year long-term exposure was carried out on S355 GP carbon steel and Z275 galvanised steel specimens in 0-4 mm sand, at four distinct moisture levels, under coupled and uncoupled conditions, in order to assess the associated mass losses and localised corrosion.
Key results
The study shows that soil moisture level is a determining parameter for the corrosion potential of both materials, owing to its control of oxygen diffusion and the active surface area. For bare carbon steel crossing the dry/saturated soil interface, DACCs induce a redistribution of corrosion kinetics without an overall aggravating effect, with the more aerated zone remaining the most corroded. However, for galvanised steel, a critical situation is identified at the interface with the water table, where accelerated consumption of the zinc coating can be observed. Despite this localised weakness, the zinc coating remains globally beneficial by reducing the localised corrosion of the underlying steel. These results also highlight the potentially aggravating role of DACCs for structures whose organic coating presents localised defects.